| Issue |
J Extra Corpor Technol
Volume 57, Number 3, September 2025
|
|
|---|---|---|
| Page(s) | 129 - 136 | |
| DOI | https://doi.org/10.1051/ject/2025006 | |
| Published online | 15 September 2025 | |
Original Article
Is hyperoxia during veno-arterial extracorporeal life support due to cardiopulmonary failure associated with mortality in pediatric patients?
1
Department of Pediatrics, Division of Cardiology, Emory University School of Medicine, Children’s Healthcare of Atlanta, Atlanta, GA 30329, USA
2
Emory University School of Medicine, Atlanta, GA 30329, USA
3
Physician Assistant, Children’s Healthcare of Atlanta, Atlanta, GA 30329, USA
4
Senior Research Coordinator, Children’s Healthcare of Atlanta, Atlanta, GA 30329, USA
5
Advanced Technology Coordinator, ECMO and Advanced Technologies, Children’s Healthcare of Atlanta, Atlanta, GA 30329, USA
6
Department of Surgery, Division of Cardiothoracic Surgery, Emory University School of Medicine, Children’s Healthcare of Atlanta, Atlanta, GA 30329, USA
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
; This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
16
July
2024
Accepted:
27
February
2025
Abstract
Background: Data is limited regarding the effects of supraphysiologic blood oxygen tension in patients requiring extracorporeal life support (ECLS). We sought to evaluate the association between hyperoxia and outcomes in pediatric patients requiring veno-arterial (VA) ECLS. Methods: Retrospective single-center study at an academic children’s hospital that included all patients 0–18 years who required VA-ECLS between 01/2014 and 12/2019. Results: During the study period, 229 VA-ECLS runs occurred in 229 patients. The majority of patients were neonates (73.4%), with cardiac being the most common indication (48.9%). The median time from admission to cannulation was 78.5 h (IQR 14, 356) with a median ECLS duration of 111.5 h (IQR 65.5, 184.5). The overall mortality rate was 44.5%. Using a receiver operating curve, a mean PaO2 of 233 mmHg in the first 48 h of ECLS was determined to have the optimal discriminatory ability for mortality (sensitivity 36% and specificity 76%). Of the VA-ECLS cohort, 68 (29.7%) had a mean PaO2 > 233 mmHg (hyperoxia group). The hyperoxia group tended to be older (median age 4.6 vs 1.5 months, p = 0.019), had a primary cardiac indication for VA-ECLS (60% vs 44%, p = 0.0004), and had a higher mortality rate (54% vs 40%, p = 0.050). In the multivariable analysis, after adjusting for covariables, the data demonstrated increased odds of mortality (aOR 2.02, 95% CI [1.03, 3.97], p = 0.03). The odds of development of stage II or III acute kidney injury (AKI) (aOR 2.04, 95% CI [0.82, 5.50]), but that did not reach statistical significance (p = 0.120). Conclusion: There is evidence that hyperoxia during the first 48 h of VA-ECLS may be associated with mortality and development of acute kidney injury, although this did not reach statistical significance. Multicenter and prospective evaluation of this modifiable risk factor is imperative to improve the care of this high-risk cohort.
Key words: Extracorporeal Life Support (ECLS) / Hyperoxia / Veno-arterial Extracorporeal Life Support (VA-ECLS) / Mortality / Functional Status Scale (FSS)
© The Author(s), published by EDP Sciences, 2025
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Extracorporeal life support (ECLS) is commonly used to support patients with reversible cardiopulmonary failure refractory to conventional medical treatment. While the primary purpose of ECLS is bridge-to-recovery, it has also been deployed as a bridge-to-bridge, bridge-to-transplantation, or bridge-to-decision [1, 2]. Since the introduction of ECLS in the mid-1970s, its utilization to support patients with complex pathologies has steadily risen. According to the most recent ELSO registry report in 2024, over 89,000 neonatal and pediatric patients have been supported with ECLS, with an overall survival rate of about 53% [3].
During veno-arterial (VA-) ECLS, deoxygenated blood is drained from the venous side of the circulation, oxygenated, and pumped back to the arterial side. ECLS circuits utilize a highly efficient oxygenator whereby the arterial blood returning to the body contains high partial pressure of oxygen (PaO2) that can exceed 400–500 mmHg. When cells are exposed to supraphysiologic levels of oxygen in the bloodstream, the term hyperoxia is used. Hyperoxia has been well studied in various clinical scenarios, including after resuscitation from cardiac arrest (CA), perinatal asphyxia, myocardial infarction, traumatic brain injury, and following cardiopulmonary bypass (CPB). Several studies in both adults and children have demonstrated an association between hyperoxia exposure and increased morbidity and mortality [2, 4–14]. Although the association of hyperoxia with adverse outcomes has been shown previously, the level at which PaO2 becomes deleterious may differ depending on the clinical situation, including the duration of exposure, the patient’s age, underlying physiology, and disease process [4, 5, 9, 13–17].
Given the lack of a clear definition of hyperoxia, we sought to evaluate hyperoxia exposure in a high-risk patient population who required ECLS for cardiopulmonary failure. Our primary aim was to determine if hyperoxia while on ECLS was associated with mortality using a derived cut-point within our cohort. Our secondary aim was to determine if hyperoxia during ECLS was associated with greater odds of new morbidity including Functional Status Scale (FSS), and the development of complications while on ECLS, such as acute kidney injury (AKI).
Materials and methods
This is a single-center retrospective cohort study in a high-volume ECLS center. All patients who required VA-ECLS due to cardiopulmonary failure between January 1, 2014, and December 31, 2019, at Children’s Healthcare of Atlanta (CHOA), a free-standing, university-affiliated quaternary children’s hospital. An internal ECLS database was queried, and eligible patient encounters were identified. The study was approved by the CHOA Institutional Review Board (IRB# 00001239, approval date: 10/11/2022). Informed consent was waived.
Data and definitions
All consecutive patients who required VA-ECLS support in index hospitalization were included. Demographic features, clinical characteristics, and ECLS variables were collected. All arterial blood gases were obtained from the patient’s arterial line during the first 48 h while on ECLS. The primary outcome was defined as all-cause ECLS mortality. The secondary outcome variables included FSS, AKI (Stage II or Stage III, as defined by the KDIGO criteria) [18] and major complications. Major complications were defined as the presence of either cardiovascular, renal or mechanical complications.
Functional Status Scale (FSS)
The FSS consists of six main domains: mental status, sensory, communication, motor function, feeding, and respiratory. Functional status for each domain was categorized from a normal score of 1 to very severe dysfunction with a score of 5, giving total FSS scores ranging from 6 to 30 as previously described [19]. Functional status scoring for this study involved retrospectively scoring baseline status (prior to admission functional status according to caregivers) and again at hospital discharge by examining the detailed history and physical exam performed by the primary medical team. FSS score determination was blinded from hyperoxia status. Newborns who had never achieved a stable baseline FSS were assigned a score of 6. This was operationalized by assigning a baseline FSS score of 6 to all admissions for infants 0–2 days old and to transfers from another facility for infants 3–6 days old, as previously reported [20–23]. New morbidity was defined as an increase in total FSS score of ≥3 points, and unfavorable functional outcomes were defined as an increase of ≥5 [24].
Clinical management
All circuits were blood primed before the start of ECLS with packed red blood cells, 25% albumin, sodium bicarbonate, calcium gluconate, and heparin for patients <40 kg. It is common practice for ABGs to be obtained at the discretion of the clinical team, most typically 30 min after initial ECLS-cannulation, and then hourly for the first 3 h. Subsequently, they are typically obtained every 3–6 h and 30 min after an adjustment in ECLS support. Target gas exchange parameters are not dictated by protocol at our center. Goal PaO2 ranges have not established as normal and the variation we describe is derived from measurements occurring during clinical care. Goal PaCO2 was 35–45 mmHg, and goal pH was 7.35–7.45. Once patients are placed on ECLS, the ventilator is placed on “rest settings” of the following: ventilator mode pressure control, peak inspiratory pressure 20 cm H2O, peak end-expiratory pressure 10 cm H2O, respiratory rate 20/minute, inspiratory time 1 second, and FiO2 30%.
Statistical analysis
Statistical analysis was conducted using SAS version 9.0 software, with a significance level set at p < 0.05. The diagnostic utility of mean PaO2 in predicting mortality was evaluated using Youden’s index (J = sensitivity + specificity − 1) and receiver operating characteristic (ROC) curves. The study population was stratified into hyperoxia and non-hyperoxia groups based on the optimal cut-off value for mean PaO2, determined by maximizing the J-value. Fisher’s exact test was employed for comparing categorical variables, while Student’s t-test and the nonparametric Wilcoxon rank-sum test were used for continuous variables, as appropriate. Additionally, a scatterplot was generated to examine the relationship between mean PaO2, duration of ECLS run, and survival, with Spearman’s correlation coefficient reported. To assess the impact of hyperoxia on mortality and AKI, univariable and multivariable logistic regression analyses were performed, adjusting for BSA, age group, and indication for ECLS in the multivariable analysis, which were determined a priori. The results are presented as odds ratios (OR) with corresponding 95% confidence intervals (CI).
Results
During the study period, 229 VA-ECLS runs occurred in 229 patients. The median age of this cohort was 2.5 months (IQR 0.3, 19.0) with a weight of 4.4 kg (IQR 3.2, 10.7), and an even distribution of males (49.8%) and females (50.2%). The majority were neonates (73.4%), with primary cardiac failure being the most common indication (48.9%) for ECLS. The median time from admission to cannulation was 78.5 h (IQR 14, 356) with a median run duration of 111.5 h (IQR 65.5, 184.5). The overall mortality rate was 44.5% (Table 1). There was a total of 3664 PaO2 samples obtained for the entire cohort. The range of samples per patient was 11–32, and the median number of samples per patient was 17 (IQR 14, 21).
Patient demographics and clinical characteristics of entire VA-ECLS cohort stratified by mean PaO2 levels in the first 48 h into non-hyperoxia group (PaO2 ≤ 233 mmHg) hyperoxia group (PaO2 > 233 mmHg).
Cut-point analysis
Using ROC analysis, PaO2 > 233 mmHg had the optimal discriminatory ability for mortality with a sensitivity of 36%, and specificity of 76%, and was defined as hyperoxia for this study population (Fig. 1). AUC for PaO2 to predict mortality was 0.55 (95% CI [0.47–0.62]; p = 0.212). Approximately one-third of the patients were in the hyperoxia group (68/229) with the remainder in the non-hyperoxia group (161/229) (Fig. 2). The two cohorts were similar in demographic variables [i.e., weight, height, body surface area (BSA), sex, and race/ethnicity] except for age (Table 1). Patients in the hyperoxia group were older [4.6 months (IQR 0.4, 27.8) vs 1.5 months (IQR 0.2, 17.2), p = 0.019] and had higher median initial ECLS flow in the first 2 h [128 mL/kg/min (IQR 97, 131) vs 109 mL/kg/min (IQR 100, 121), p = 0.018]. The most common indication to require ECLS (based on ELSO registry criteria) was cardiac in both groups (60.3% and 44.1%, respectively). Patients in the hyperoxia group had a lower rate of expected recovery and ability to wean off ECLS (54.4% vs 73%, p = 0.049). Compared to the non-hyperoxia group, we found that mortality was higher in the hyperoxia group (54.4% vs 40.45, p = 0.050) (Table 1, Fig. 2).
 |
Figure 1 Receiver operating characteristic (ROC) curve identifying the optimal discriminatory cut point for mortality was 233 mmHg (sensitivity 36%, specificity 76%). |
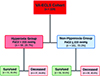 |
Figure 2 Flow chart of neonates requiring Veno-arterial Extracorporeal Life Support (VA-ECLS) stratified based on PaO2 levels in the first 48 h while on ECLS. |
Outcomes analysis
In the univariable analysis, we found that the hyperoxia group had increased odds of mortality (OR 1.76, 95% CI [0.995–3.12], p = 0.052). In the multivariable analysis when controlling for age group (neonates vs pediatrics) and indication for ECLS, hyperoxia continued to demonstrate higher odds of mortality (aOR 2.02, 95% CI [1.03–3.97], p = 0.03) and higher odds of developing stage II or III AKI (aOR 2.12 95% CI [0.82–5.50], p = 0.12) (Table 2), but this did not reach statistical significance. The association of average PaO2 and ECLS duration is graphically demonstrated in Figure 3 (correlation coefficient −0.11, 95% CI [−0.33 – 0.03], p = 0.093).
 |
Figure 3 Scatterplot illustrating the relationship of average PaO2, VA-ECLS run duration, and mortality in the VA-ECLS Cohort. |
Outcomes of patients undergoing VA-ECLS using a univariable and multivariable regression analysis.
Functional Status Scale (FSS) of survivors and development of new morbidity and unfavorable outcomes
Among the non-hyperoxia group, 21 of the 96 survivors (21.9%) developed new morbidity along with 9 out of 31 survivors (29.0%) of the hyperoxia group. Unfavorable functional outcome (defined as an increase in FSS ≥ 5) occurred in 8/96 patients in the non-hyperoxia survivors (8.3%), and 2/31 (6.5%) in the non-hyperoxia group. We failed to identify an association between designation as “hyperoxia” and new morbidity or unfavorable outcomes (Table 3).
New morbidity and unfavorable functional outcome for overall survivors who required VA-ECLS stratified by PaO2 levels into hyperoxia and non-hyperoxia groups based on functional status scale change from admission to discharge.
Subset analysis of patients with a cardiac diagnosis who required ECLS post-cardiotomy
There were a total of 196 patients with a cardiac diagnosis who required VA-ECLS. Of those, 155 were post-cardiotomy and are shown in Supplemental Table 1. We stratified the post-cardiotomy patients using the PaO2 cut point that was used above (PaO2 233 mmHg) and found that patients in the hyperoxia group had a longer duration of ECLS run (149 vs 88 h, p = 0.0004), and had a lower rates of expected recovery (66.1 vs 78.1, p = 0.027). The full comparison between the cohorts is shown in Supplemental Table 2.
Discussion
In this single-center retrospective report from a high-volume ECLS center, we describe an overall mortality rate of patients supported on VA-ECLS of 44.5%. Using a ROC curve, a mean PaO2 of 233 mmHg in the first 48 h of ECLS was determined to have the optimal discriminatory ability for mortality (sensitivity 36% and specificity 76%). Of the VA-ECLS runs, 68 (29.7%) had mean PaO2 > 233 mmHg and were categorized as the hyperoxia group. Patients in the hyperoxia group tended to be older (median age 4.6 vs 1.5 months, p = 0.019), to have a cardiac indication for VA-ECLS (60% vs 44%, p = 0.0004), and had lower rates of expected recovery to come off ECLS (54% vs 73%, p = 0.049). Multi-variable logistic regression controlling for confounding variables demonstrated a suggestive association, though it failed to reach statistical significance, between hyperoxia and mortality in the unadjusted analysis (OR 1.76, 95% CI [0.995–3.12], p = 0.052). While hyperoxia during ECLS did not directly lead to death, we postulate that it may contribute to new morbidities, such as acute kidney injury, that would later lead to complications and mortality.
In other critical illness settings, an association between excessive oxygen delivery and poor clinical outcomes has been reported. In patients requiring ECLS post-cardiac arrest, hyperoxia (as defined by a mean PaO2 > 193 mmHg) was associated with 30-day mortality and the need for dialysis [4, 15, 25]. In a large multicenter cohort study of adult patients admitted to the ICU after resuscitation from cardiac arrest (CA), Kilgannon et al. showed an association between hyperoxia and risk of in-hospital death consistent with a dose-dependent relationship [15]. In a prospective disease-specific CA database, Elmer and colleagues found that exposure to severe hyperoxia was independently associated with inpatient mortality [25]. A retrospective study by Al-Kawaz and colleagues demonstrated poor neurological outcomes in patients exposed to mild, moderate, and severe hyperoxia in the first 24 h of ECLS. Additional duration of severe hyperoxia was independently associated with in-hospital mortality [13]. Several reports of neonates with asphyxia have demonstrated an association between hyperoxia and an increased risk of brain injury and mortality [4, 26, 27]. Conversely, Raman et al, in a single-center study and systematic review of a heterogeneous cohort of critically ill patients, did not demonstrate an association between hyperoxia at the time of admission and mortality [28].
In a prior report, we showed that a substantial portion of infants undergoing cardiac surgery using CPB were exposed to hyperoxia and that patients in the hyperoxia group had four-fold greater odds of mortality within 30 days of surgery [9]. This report supports earlier findings that hyperoxia is likely associated with worse outcomes, but which populations are at risk remains unclear, and the impact of other clinical variables that may affect oxygenation in a direct or indirect way. Some of these factors are patient hemoglobin levels, ventilator settings including FiO2, the health and age of the oxygenator in the ECLS circuit, ECLS flows, recirculation, and whether the patient is sedated and paralyzed to decrease oxygen consumption. It would be extremely useful to control these factors, but in reality, the degree of impact of each factor is different for each patient. This really supports the importance of this study and future studies to help understand the true impact of oxygen on patient outcomes and on the biological systems of the body. However, the delineation of risk categories in different patient populations remains unclear.
There is no generally accepted value to discriminate pathologic hyperoxia. Injurious hyperoxia may vary by patient population and clinical context [25]. Poor outcomes may occur when PaO2 exceeds a certain threshold, where endogenous antioxidants are unable to prevent oxidative stress. When high amounts of oxygen are introduced to previously ischemic tissues, this leads to the generation of reactive oxygen species (ROS) and activation of inflammatory pathways via cytokines and other immunological signaling pathways [29]. The generation of ROS leads to lipid peroxidation and protein changes, resulting in cell injury. In patients who have experienced CA or resuscitation aftershock, an increase in ROS may deplete plasma antioxidant potential, which may lower the threshold for subsequent oxidative injury [14, 25], and this effect may be more pronounced in neonates and infants due to immature antioxidant defenses [4]. The effect of relative hyperoxia may be even more pronounced in patients with cyanotic heart disease who have significantly lower baseline PaO2. It is not known if the antioxidant systems of the body are downregulated in patients with chronically lower baseline PaO2. When placed on ECLS, these patients are exposed to a relative hyperoxia state. It is plausible that these patients are more vulnerable to supraphysiologic oxygen, the extent of which is yet unknown.
Because there is no generally accepted definition of hyperoxia in neonates with congenital heart disease with various physiology and complexity in different settings, we used a ROC curve analysis in this specific cohort to determine which PaO2 values may be associated with adverse outcomes. This similar strategy was employed by Sznycer-Taub et al., and Beshish et al. in two separate reports. Sznycer-Taub and colleagues evaluated hyperoxia in pediatric cardiac patients (neonates and infants) supported on VA-ECLS and found that a PaO2 of 193 mmHg in the first 48 h was determined to have good discriminatory ability about 30-day mortality [4]. Using a similar strategy, Beshish and colleagues showed that a PaO2 of 313 mmHg for infants undergoing cardiac surgery utilizing CPB was independently associated with 30-day mortality [9]. Our cut-off definition of hyperoxia was very close to that identified by Sznycer-Taub et al. although the patient population that was used by Sznycer-Taub was slightly different, as the former study captured all infants who required VA-ECLS following cardiac surgery. In that study, the cutoff point remained the same (PaO2 of 193 mmHg) in a subgroup analysis of all neonates (n = 70), and in neonates who underwent a Norwood operation (n = 35).
Limitations
Our findings are subject to all limitations inherent to single-center retrospective cohort studies. Although samples to measure PaO2 were obtained at dedicated time intervals, it is not possible to discern the effect of time spent in a hyperoxia state as opposed to the effects of acutely high PaO2 levels. Additionally, there may be some bias as to which patients are exposed to hyperoxia. The majority of our cohort had a PaO2 level around or over 200 mmHg while on VA-ECLS, which limited our ability to study the relationship between lower oxygen tension levels and outcomes. Although we identified a cut point for PaO2 of 233 mmHg using an AUC, the sensitivity was 36%. The sensitivity is low, and this is clearly a limitation of our study that we think can be overcome with a larger patient population that is more homogenous. Importantly, many of these limitations can be addressed in a multicenter validation study, which our group is currently pursuing. It is acceptable that the AUC for the model is low, which to the complexity of hyperoxia, which might not be properly captured by the variables in the model. There is also the possibility of interaction between predictors, which was not accounted for. An alternative way that can be done in future analyses is to combine different sets of predictors or explore other analytic approaches that might be able to yield strong associations within certain patient populations. Also, when presenting findings in Figure 3, we show that the correlation between PaO2 level, duration of ECLS and mortality was not significant. This could be related to many factors such as the heterogeneous patient population, patient response to ECLS exposure, severity of illness, and other Confounding factors, which can also play a crucial role, such that the important predictors might not have been accounted for in the correlation, to name a few.
Conclusions
Of the 229 VA-ECLS runs in 209 patients, hyperoxia during the first 48 h of VA-ECLS defined by a mean PaO2 > 233 mmHg, occurred in approximately 30% of runs. Patients in the hyperoxia group were older and had higher median initial ECLS flow in the first 2 h. The mortality rate was higher in the hyperoxia group, and although it did not reach statistical significance, patients in the hyperoxia group had higher odds of mortality (p = 0.052). Multicenter and prospective evaluation of this modifiable risk factor is imperative to improve the care of this high-risk cohort.
Funding
This research did not receive any specific funding.
Conflicts of interest
The authors declared no conflict of interest.
Data availability statement
All available data are incorporated into the article.
Author contribution statement
A.B. and H.V. designed the study. P.R.-M., R.S., J.Q., K.K.-L., T.Z., and A.B. performed the research and analyzed the data. A.B. wrote the manuscript, and all authors contributed to the final version.
Ethics approval
Data collection was conducted as a retrospective cohort study to determine the ranges of PaO2 exposure and the potential association between exposure to hyperoxia and poor outcomes. Our primary aim was to determine if hyperoxia while on VA-ECLS was associated with increased mortality using a derived cut-point within our cohort. Our secondary aim was to determine if hyperoxia during VA-ECLS is associated with greater odds of morbidity using the Functional Status Scale (FSS), and the development of complications while on ECLS, including acute kidney injury (AKI). The study protocol conformed to the Declaration of Helsinki and was approved by the institution’s ethics committee in Children’s Healthcare of Atlanta, Atlanta, Georgia, USA. Medical research is subject to ethical standards that promote and ensure respect for all human subjects and protect their health and rights.
This study was approved by the Children’s Healthcare of Atlanta Institutional Review Board: Study ID No. (IRB# 00001239), approved on 10/11/2022.
Supplementary material
Supplemental Table 1. Cardiac Diagnosis of Patient in the VA-ECLS Cohort.
Supplemental Table 2. Patient Demographics and Clinical Characteristics of Patients Who Required VA-ECLS Post-Cardiotomy Stratified by Mean PaO2 Levels in the First 48 h into Non-Hyperoxia Group (PaO2 ≤ 233 mmHg), Hyperoxia Group (PaO2 > 233 mmHg).
Access Supplementary MaterialReferences
- Munshi L, Kiss A, Cypel M, Keshavjee S, Ferguson ND, Fan E. Oxygen thresholds and mortality during extracorporeal life support in adult patients. Crit Care Med. 2017;45(12):1997–2005. [CrossRef] [PubMed] [Google Scholar]
- Raffaeli G, Ghirardello S, Passera S, Mosca F, Cavallaro G. Oxidative stress and neonatal respiratory extracorporeal membrane oxygenation. Front Physiol. 2018;9:1739. [CrossRef] [PubMed] [Google Scholar]
- ELSO. Registry of the extracorporeal life support organization. Ann Arbor ME: Registry Report; 2024. [Google Scholar]
- Sznycer-Taub NR, Lowery R, Yu S, Owens ST, Hirsch-Romano JC, Owens GE. Hyperoxia is associated with poor outcomes in pediatric cardiac patients supported on venoarterial extracorporeal membrane oxygenation. Pediatr Crit Care Med. 2016;17(4):350–358. [CrossRef] [PubMed] [Google Scholar]
- Cashen K, Reeder R, Dalton HJ, et al. Hyperoxia and hypocapnia during pediatric extracorporeal membrane oxygenation: associations with complications, mortality, and functional status among survivors. Pediatr Crit Care Med. 2018;19(3):245–253. [CrossRef] [PubMed] [Google Scholar]
- Ni YN, Wang YM, Liang BM, Liang ZA. The effect of hyperoxia on mortality in critically ill patients: a systematic review and meta analysis. BMC Pulm Med. 2019;19(1):53. [CrossRef] [PubMed] [Google Scholar]
- Bonnemain J, Rusca M, Ltaief Z, et al. Hyperoxia during extracorporeal cardiopulmonary resuscitation for refractory cardiac arrest is associated with severe circulatory failure and increased mortality. BMC Cardiovasc Disord. 2021;21(1):542. [CrossRef] [PubMed] [Google Scholar]
- Brown DM, Holt DW, Edwards JT, Burnett RJ. 3rd: Normoxia vs. hyperoxia: impact of oxygen tension strategies on outcomes for patients receiving cardiopulmonary bypass for routine cardiac surgical repair. J Extra Corpor Technol. 2006;38(3):241–248. [CrossRef] [EDP Sciences] [PubMed] [Google Scholar]
- Beshish AG, Jahadi O, Mello A, Yarlagadda VV, Shin AY, Kwiatkowski DM. Hyperoxia during cardiopulmonary bypass is associated with mortality in infants undergoing cardiac surgery. Pediatr Crit Care Med. 2021;22(5):445–453. [CrossRef] [PubMed] [Google Scholar]
- Lilien TA, Groeneveld NS, van Etten-Jamaludin F, et al. Association of arterial hyperoxia with outcomes in critically Ill children: a systematic review and meta-analysis. JAMA Netw Open. 2022; 5(1):e2142105. [CrossRef] [PubMed] [Google Scholar]
- Stoll SE, Paul E, Pilcher D, Udy A, Burrell A. Hyperoxia and mortality in conventional versus extracorporeal cardiopulmonary resuscitation. J Crit Care. 2022;69:154001. [CrossRef] [PubMed] [Google Scholar]
- McDonald CI, Fraser JF, Coombes JS, Fung YL. Oxidative stress during extracorporeal circulation. Eur J Cardiothorac Surg. 2014;46(6):937–943. [CrossRef] [PubMed] [Google Scholar]
- Al-Kawaz MN, Canner J, Caturegli G, et al. Duration of hyperoxia and neurologic outcomes in patients undergoing extracorporeal membrane oxygenation. Crit Care Med. 2021;49(10):e968–e977. [CrossRef] [PubMed] [Google Scholar]
- Chang WT, Wang CH, Lai CH, et al. Optimal arterial blood oxygen tension in the early postresuscitation phase of extracorporeal cardiopulmonary resuscitation: A 15-year retrospective observational study. Crit Care Med. 2019;47(11):1549–1556. [CrossRef] [PubMed] [Google Scholar]
- Kilgannon JH, Jones AE, Parrillo JE, et al. Relationship between supranormal oxygen tension and outcome after resuscitation from cardiac arrest. Circulation. 2011;123(23):2717–2722. [CrossRef] [PubMed] [Google Scholar]
- Page D, Ablordeppey E, Wessman BT, et al. Emergency department hyperoxia is associated with increased mortality in mechanically ventilated patients: a cohort study. Crit Care. 2018;22(1):9. [CrossRef] [PubMed] [Google Scholar]
- Davis DP, Meade W, Sise MJ, et al. Both hypoxemia and extreme hyperoxemia may be detrimental in patients with severe traumatic brain injury. J Neurotrauma. 2009;26(12):2217–2223. [CrossRef] [PubMed] [Google Scholar]
- Stevens PE, Levin A. Evaluation and management of chronic kidney disease: synopsis of the kidney disease: improving global outcomes 2012 clinical practice guideline. Ann Intern Med. 2013;158(11):825–830. [CrossRef] [PubMed] [Google Scholar]
- Pollack MM, Holubkov R, Glass P, et al. Functional Status Scale: new pediatric outcome measure. Pediatrics. 2009;124(1):e18–e28. [CrossRef] [PubMed] [Google Scholar]
- Berg RA, Nadkarni VM, Clark AE, et al. Incidence and outcomes of cardiopulmonary resuscitation in PICUs. Crit Care Med. 2016;44(4):798–808. [PubMed] [Google Scholar]
- Beshish AG, Baginski MR, Johnson TJ, Deatrick BK, Barbaro RP, Owens GE. Functional status change among children with extracorporeal membrane oxygenation to support cardiopulmonary resuscitation in a pediatric cardiac ICU: a single institution report. Pediatr Crit Care Med. 2018;19(7):665–671. [CrossRef] [PubMed] [Google Scholar]
- Beshish AG, Rodriguez Z, Hani Farhat M, et al. Functional status change among infants, children, and adolescents following extracorporeal life support: a multicenter report. ASAIO J. 2022;69(1):114–121. [Google Scholar]
- Han B, Yang JK, Ling AY, et al. Early functional status after surgery for congenital heart disease: a single-center retrospective study. Pediatr Crit Care Med. 2021;23(2):109–117. [Google Scholar]
- Pollack MM, Holubkov R, Funai T, et al. Relationship between the functional status scale and the pediatric overall performance category and pediatric cerebral performance category scales. JAMA Pediatr. 2014;168(7):671–676. [CrossRef] [PubMed] [Google Scholar]
- Elmer J, Scutella M, Pullalarevu R, et al. The association between hyperoxia and patient outcomes after cardiac arrest: analysis of a high-resolution database. Intensive Care Med. 2015;41(1):49–57. [CrossRef] [PubMed] [Google Scholar]
- Klinger G, Beyene J, Shah P, Perlman M. Do hyperoxaemia and hypocapnia add to the risk of brain injury after intrapartum asphyxia? Arch Dis Child Fetal Neonatal Ed. 2005;90(1):49–52. [Google Scholar]
- Rabi Y, Rabi D, Yee W. Room air resuscitation of the depressed newborn: a systematic review and meta-analysis. Resuscitation. 2007;72(3):353–363. [CrossRef] [PubMed] [Google Scholar]
- Raman S, Prince NJ, Hoskote A, Peters MJ. Admission PaO2 and mortality in critically ill children: a cohort study and systematic review. Pediatr Crit Care Med. 2016;17(10):e444–e450. [CrossRef] [PubMed] [Google Scholar]
- Turer AT, Hill JA. Pathogenesis of myocardial ischemia-reperfusion injury and rationale for therapy. Am J Cardiol. 2010;106(3):360–368. [CrossRef] [PubMed] [Google Scholar]
Cite this article as: Beshish AG, Shamah R, Qian J, Keane-Lerner K, Morales PR, Zinyandu T, Davis J, Rosenblum JM & Viamonte HK. Is hyperoxia during veno-arterial extracorporeal life support due to cardiopulmonary failure associated with mortality in pediatric patients? J Extra Corpor Technol 2025, 57, 129–136. https://doi.org/10.1051/ject/2025006.
All Tables
Patient demographics and clinical characteristics of entire VA-ECLS cohort stratified by mean PaO2 levels in the first 48 h into non-hyperoxia group (PaO2 ≤ 233 mmHg) hyperoxia group (PaO2 > 233 mmHg).
Outcomes of patients undergoing VA-ECLS using a univariable and multivariable regression analysis.
New morbidity and unfavorable functional outcome for overall survivors who required VA-ECLS stratified by PaO2 levels into hyperoxia and non-hyperoxia groups based on functional status scale change from admission to discharge.
All Figures
 |
Figure 1 Receiver operating characteristic (ROC) curve identifying the optimal discriminatory cut point for mortality was 233 mmHg (sensitivity 36%, specificity 76%). |
| In the text | |
 |
Figure 2 Flow chart of neonates requiring Veno-arterial Extracorporeal Life Support (VA-ECLS) stratified based on PaO2 levels in the first 48 h while on ECLS. |
| In the text | |
 |
Figure 3 Scatterplot illustrating the relationship of average PaO2, VA-ECLS run duration, and mortality in the VA-ECLS Cohort. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


