| Issue |
J Extra Corpor Technol
Volume 58, Number 1, March 2026
|
|
|---|---|---|
| Page(s) | 39 - 42 | |
| DOI | https://doi.org/10.1051/ject/2025061 | |
| Published online | 13 March 2026 | |
Original Article
Incidence and pathophysiology of gastrointestinal bleeding during mechanical circulatory support: A retrospective analysis using machine learning algorithms
1
Department of Cardiovascular Perfusion and Extracorporeal Technology, Ochsner Health, 1514 Jefferson Highway, New Orleans, Louisiana 70121, USA
2
CES University, Cl 10A #22-04, El Poblado, Medellin, Antioquia, Columbia
3
The University of Queensland Medical School, 288 Herston Road, Herston, QLD 4006, Australia
4
Critical Care Section, Anesthesiology & Perioperative Medicine, Ochsner Health, 1514 Jefferson Highway, New Orleans, Louisiana 70121, USA
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
7
April
2025
Accepted:
12
October
2025
Abstract
Background: End-organ hypoperfusion from cardiopulmonary shock may require mechanical circulatory support (MCS). However, patients receiving MCS risk the development of hemorrhagic complications, including gastrointestinal bleeding (GI). Examining potential risk factors for these complications improves clinical understanding. The purpose of this investigation was to study the risk for GI bleeding in MCS patients. Methods: Following IRB approval, patient characteristics, previously reported comorbidities, and the incidence of GI bleeding were reviewed from January 2017 to October 2023. Clinical variables underwent machine learning with autovalidation. Support vector machine modeling provided the best performance among the ensemble models tested. Results: In this study of 156 patients who underwent 284 MCS procedures, the incidence of GI bleeding was 6.0% CI 3.3–10.4%. Following machine learning, patients with insulin-dependent diabetes were associated with GI bleeding. The Receiver Operating Characteristic (ROC) curve demonstrated an area under the curve (AUC) of 0.85 with a misclassification rate of 7.5%. The relative risk of the need for major transfusion (>2 packed red blood cell units/episode) was 1.7 CI 1.1–2.5. The majority (87%), but not all, of these patients received unfractionated heparin therapy. Finally, hospital length of stay was increased in patients with GI bleeding. Conclusion: Insulin-dependent diabetes was associated with increased risk for GI bleeding during MCS, and these patients more often required major transfusions. Further evaluation of continuous anticoagulation therapy is warranted. Knowledge derived from this analytical study may guide the development of institutional protocols to improve care in this patient population.
Key words: Gastrointestinal bleeding / Mechanical circulatory support / Transfusion / Hemorrhage / Insulin-dependent diabetes
© The Author(s), published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Cardiopulmonary shock (CS) is characterized by low cardiac output and profound hypotension that can result in end-organ failure [1–4]. Patients refractory to conventional therapies, such as inotropes, vasopressors, and/or mechanical ventilation, may require mechanical circulatory support (MCS) to restore perfusion [1–5]. MCS improves deranged hemodynamics and restores perfusion to the organs. However, MCS is not without complications such as bleeding, thrombosis, and hemolysis [4–7]. The risk of bleeding is exacerbated by the administration of systemic anticoagulation medications commonly given after MCS initiation, such as unfractionated heparin [5, 6, 8].
Gastrointestinal (GI) bleeding is a recognized complication of MCS [5–9]. Its pathophysiology is multifactorial. Shear stress from MCS disrupts platelet function and impairs von Willebrand factor (vWF), predisposing to acquired von Willebrand syndrome (aVWS) [5, 10]. Low pulsatility during MCS may also cause mucosal hypoperfusion, ischemia, angiodysplasia, and eventual GI bleeding [5, 6, 9, 11]. In normal physiology, pulsatile flow regulates nitric oxide (NO) release from endothelial cells. With reduced pulsatility during MCS, impaired NO release can worsen hypotension and GI hypoperfusion [5].
This complex relationship between MCS physiology and GI bleeding warrants further investigation. Therefore, the purpose of this analytical study was to examine the preprocedural risk factors for GI bleeding in patients receiving MCS.
Material and methods
Following IRB approval to allow electronic health records review without prior consent, data were entered into a study database. We conducted a retrospective single-center study of 156 patients who received 284 MCS devices (Appendix, Table A1) between January 2017 and October 2023 at Ochsner Health – Jefferson Highway Campus, New Orleans, Louisiana. Patients ≥18 years were included. No exclusion criteria were applied.
Statistics
Preprocedural comorbidities and patient characteristics (Table 1) underwent machine learning with autovalidation. Model performance was assessed using AUC and misclassification rates. Support Vector Machine modeling provided the best fit. JMP Pro 18.2 was used for all statistical analyses.
Baseline characteristics and previously reported co-morbidities for gastrointestinal bleeding in 156 patients requiring mechanical circulatory support.
Results
The incidence of GI bleeding was 6.0% (CI 3.3–10.4%). Insulin-dependent diabetes was associated with increased risk of GI bleeding (Table 1). Our institution applies insulin-guided protocols for glycemic management before and during MCS. The ROC curve for the Support Vector Machine model (Appendix, Figure A1) demonstrated an AUC of 0.85 and a misclassification rate of 7.5%. The curve shows sensitivity versus 1-specificity. The steep initial rise indicates strong discriminative power, and the AUC demonstrates performance substantially above random (grey diagonal). The step-like pattern reflects limited data, highlighting the need for validation in larger cohorts. Patients with GI bleeding were more likely to require major transfusion (>2 units pRBC/episode), with a relative risk of 1.7 (CI 1.1–2.5). Most (87%) received unfractionated heparin therapy in accordance with institutional guidelines. GI bleeding was also associated with prolonged hospital length of stay (Figure 1).
 |
Figure 1 Hospital length of stay in MCS patients with GI bleeding. |
Discussion
This investigation is, to our knowledge, the first report of increased GI bleeding among insulin-dependent diabetic patients receiving MCS across a range of device types. MCS confers substantial bleeding risk [6], and comorbidity profiles should guide individualized therapies. Further investigation of anticoagulation protocols, particularly continuous therapy, is warranted. GI bleeding was defined as hematochezia, hematemesis, melena, bloody NG output, transfusion requirement, or active bleeding observed at endoscopy. Patients with GI bleeding had nearly twice the relative risk of requiring major transfusion [>2 units of packed red blood cells (pRBC)]. Transfusion may control bleeding but can worsen systemic inflammation, particularly in diabetic patients with baseline mucosal vulnerability [12, 13]. Prior studies, including Kapuria et al., have demonstrated higher rebleeding rates in diabetic patients on MCS [10].
In our cohort, GI bleeding was also associated with longer hospitalization, consistent with prior studies [6]. Together, these findings support the importance of comorbidity assessment in risk stratification for MCS patients.
Limitations
This study has limitations inherent to a retrospective single-center design. Data completeness, though strengthened by electronic medical records, may be imperfect. Another limitation is the lack of device-type analysis. Our current sample size does not permit robust statistical comparisons between device types. However, all MCS devices share intravascular access to the systemic circulation, triggering inflammation and activation of the coagulation cascade – a mechanism particularly relevant in insulin-dependent diabetes. Future studies should prioritize device-specific analyses.
Conclusions
Insulin-dependent diabetes was associated with an increased risk for GI bleeding during MCS. Patients with this complication more often required major transfusions and experienced longer hospital stays. This suggests the need for further investigation into anticoagulation strategies. Knowledge derived from this analytical study may inform institutional protocols to improve outcomes in this population.
Funding
The authors received no funding to complete this research.
Conflicts of interest
The authors declare no conflict of interest.
Data availability statement
All available data are incorporated into the article.
Author contribution statement
Kelsey Gore, BS, RRT: Design, Data harvest, Editorial review of the manuscript.
Dean Linder, Jr., CCP, LP: Editorial review of the manuscript.
Juan José Martinez Duque, MD: Data harvest, Editorial review of the manuscript.
Junxi Wang BS: Data harvest, Editorial review of the manuscript.
Connor Rudnicki DO: Editorial review of the manuscript.
Shaun Yockelson, MD: Editorial review of the manuscript.
Adrian Alexis Ruiz, MD: Editorial review of the manuscript.
Bobby D. Nossaman, MD: Research Design, Statistics, Data harvest, Editorial review of the manuscript.
Ethics approval
IRB approval 2023.201- MCS
References
- Sarraf M, Vallabhajosula S, Nagaraja V. Cardiogenic shock and percutaneous left ventricular assist devices–Investigating gender-specific disparities. Heart Lung Circul. 2024;33(12):1611–1613. https://doi.org/10.1016/j.hlc.2024.11.005. [Google Scholar]
- Ostadal P, Rokyta R, Kruger A, et al. Extra corporeal membrane oxygenation in the therapy of cardiogenic shock (ECMO-CS): rationale and design of the multicenter randomized trial. Eur J Heart Fail. 2017;19(Suppl 2):124–127. https://doi.org/10.1002/ejhf.857. [Google Scholar]
- Liden H, Wiklund L, Haraldsson A, Berglin E, Hultman J, Dellgren G. Temporary circulatory support with extra corporeal membrane oxygenation in adults with refractory cardiogenic shock. Scand Cardiovasc J. 2009;43(4):226–232. https://doi.org/10.1080/14017430802596420. [Google Scholar]
- Nakata J, Yamamoto T, Saku K, et al. Mechanical circulatory support in cardiogenic shock. J Intensive Care. 2023;11:64. https://doi.org/10.1186/s40560-023-00710-2. [Google Scholar]
- Sargent CR, Ali S, Kanamarlapudi V. The evolution and complications of long-term mechanical circulatory support devices. Hearts. 2024;5(1):105–121. https://doi.org/10.3390/hearts5010008. [Google Scholar]
- Pahuja M, Mony S, Adegbala O, et al. Incidence and outcomes of gastrointestinal bleeding in patients with percutaneous mechanical circulatory support devices. Am J Cardiol. 2022;174:76–83. https://doi.org/10.1016/j.amjcard.2022.03.025. [Google Scholar]
- Abbas A, Mahmoud A, Ahmed M, et al. Gastrointestinal bleeding during the index hospitalization for mechanical circulatory support devices implantation, a nationwide perspective. Digest Diseases Sci. 2017;62(1):161–174. https://doi.org/10.1007/s10620-016-4271-6. [Google Scholar]
- Kanji R, Vandenbriele C, Arachchillage DRJ, et al. Optimal tests to minimise bleeding and ischaemic complications in patients on extracorporeal membrane oxygenation. Thromb Haemost. 2022;122(4):480–491. https://doi.org/10.1055/a-1508-8230. [CrossRef] [PubMed] [Google Scholar]
- Singh G, Albeldawi M, Kalra SS, et al. Features of patients with gastrointestinal bleeding after implantation of ventricular assist devices. Clin Gastroenterol Hepatol. 2015;13(1):107–14.e1. https://doi.org/10.1016/j.cgh.2014.05.012. [Google Scholar]
- Kapuria D, Khumri T, Shamim S, et al. Characterization and timing of gastrointestinal bleeding in continuous flow left ventricular assist device recipients. Heliyon. 2020;6(9):e04695. https://doi.org/10.1016/j.heliyon.2020.e04695. [Google Scholar]
- Aggarwal A, Pant R, Kumar S, et al. Incidence and management of gastrointestinal bleeding with continuous flow assist devices. Ann Thoracic Surg. 2012;93(5):1534–1540. https://doi.org/10.1016/j.athoracsur.2012.02.035. [Google Scholar]
- Montoro M, Cucala M, Lanas Á, et al. Indications and hemoglobin thresholds for red blood cell transfusion and iron replacement in adults with gastrointestinal bleeding: An algorithm proposed by gastroenterologists and patient blood management experts. Front Med. 2022;9:903739. https://doi.org/10.3389/fmed.2022.903739. [Google Scholar]
- Zheng S, Ma J. A systematic review of gastrointestinal manifestations in diabetic nephropathy. Explor Res Hypothesis Med. 2019;4(3):52–61. https://doi.org/10.14218/ERHM.2019.00014. [Google Scholar]
Cite this article as: Gore K, Linder D, Duque JJM, Wang J, Rudnicki C, Alexis Ruiz A, Yockelson S & Nossaman B. Incidence and pathophysiology of gastrointestinal bleeding during mechanical circulatory support: A retrospective analysis using machine learning algorithms. J Extra Corpor Technol 2026, 58, 39–42. https://doi.org/10.1051/ject/2025061.
Appendix
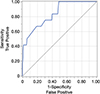 |
Figure A1 Receiver Operator Characteristic (ROC) between sensitivity and 1-specificity across classification thresholds. |
Acute and durable mechanical circulatory devices within our study cohort.
All Tables
Baseline characteristics and previously reported co-morbidities for gastrointestinal bleeding in 156 patients requiring mechanical circulatory support.
All Figures
 |
Figure 1 Hospital length of stay in MCS patients with GI bleeding. |
| In the text | |
 |
Figure A1 Receiver Operator Characteristic (ROC) between sensitivity and 1-specificity across classification thresholds. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


