| Issue |
J Extra Corpor Technol
Volume 57, Number 3, September 2025
|
|
|---|---|---|
| Page(s) | 137 - 146 | |
| DOI | https://doi.org/10.1051/ject/2025021 | |
| Published online | 15 September 2025 | |
Technique Or Application
Automated temperature management during cardiopulmonary bypass: a step toward safety and precision perfusion
1
Université, Paris-Sorbonne, 27 rue Chaligny, 75012 Paris, France
2
University of Insubria, Via Ravasi, 2, 21100 Varese, VA, Italy
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
12
March
2025
Accepted:
20
May
2025
Abstract
Precise temperature management during cardiopulmonary bypass (CPB) is crucial for optimizing patient outcomes, and influencing metabolic rate, organ protection, and neurological integrity. Traditionally, temperature control during CPB has relied on manual adjustments by perfusionists, a practice fraught with potential for human error and variability in outcomes. Such variability can lead to severe complications, including cerebral hyperthermia and inflammatory responses, which significantly impact patient recovery and morbidity. This paper introduces a novel, fully automated temperature management system, which integrates with existing heater-cooler units (HCUs) and advanced perfusion systems to enhance precision and reliability. By utilizing real-time physiological monitoring and intelligent automation, the system dynamically adjusts temperature phases based on continuous patient feedback. Preliminary simulation data are presented to validate the system’s feasibility and responsiveness. Ethical considerations regarding automated decision-making in surgery are also briefly discussed.
Key words: Cardiopulmonary bypass / Temperature management / Automated systems / Perfusion systems / Patient safety / Intelligent automation
© The Author(s), published by EDP Sciences, 2025
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Cardiopulmonary bypass (CPB) is a critical component of cardiac surgery, enabling life-saving procedures by maintaining circulatory and respiratory support when the heart and lungs must be temporarily ceased. Despite the pivotal role of CPB in modern surgery, managing the patient’s body temperature during such procedures remains a substantial challenge, with profound implications for patient outcomes [1]. Traditional temperature management during CPB relies predominantly on manual adjustments performed by perfusionists. This manual control, while experience-based, introduces a significant variability in patient outcomes, primarily due to the subjective nature of human decision-making and the potential for error. Such inconsistencies are not trivial; they are linked to serious complications, including cerebral hyperthermia, which can lead to neurological damage, and improper cooling or rewarming, which may exacerbate the inflammatory response following surgery [2]. Precise control of temperature is crucial because metabolic demands and the integrity of neurological and other organ functions are highly temperature-sensitive. Hypothermia, commonly induced during CPB, is intended to reduce metabolic rate and protect neurological functions by decreasing the oxygen needs of the brain and other critical organs [3, 4]. However, achieving and maintaining the correct degree of hypothermia, followed by a controlled rewarming phase, requires meticulous management to avoid the adverse effects of temperature fluctuations. The risks associated with suboptimal temperature control include not only neurological impairments but also coagulopathies, arrhythmias, and compromised immune functions, which collectively contribute to a complex postoperative recovery [5]. The introduction of automated systems in medical fields has consistently demonstrated enhanced outcomes through increased precision and reduced human error. In the context of CPB, an automated temperature management system could revolutionize standard practices by providing more consistent, precise, and safe temperature control [6, 7]. This paper proposes the development of such a system, integrating advanced sensor technologies and intelligent algorithms with existing perfusion and heater-cooler systems [1]. The goal is to automate the cooling and rewarming phases of CPB, leveraging real-time physiological data to dynamically adjust to the optimal temperature settings tailored to individual patient needs [8]. This innovative approach aims not only to standardize temperature management across surgical teams but also to enhance patient safety and improve clinical outcomes by reducing the variability inherent in manual processes. By discussing the integration of this technology, the paper explores its potential to set new benchmarks in cardiac surgery, emphasizing the crucial role of technology in advancing medical practice and patient care.
Materials and methods
A narrative review was conducted to support the development of the proposed algorithm. We searched PubMed with Boolean combinations of terms: “Oxygen Delivery,” “Cardiopulmonary Bypass,” “Temperature Management,” “Automated Systems,” and “Patient Safety.” The initial search yielded 152 articles. After screening abstracts and full texts using predefined inclusion criteria, 34 articles were shortlisted. A final selection of 12 core studies was made. A PRISMA-style flowchart illustrating the study selection process is shown in Figure 1.
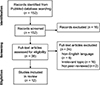 |
Figure 1 PRISMA flow chart. |
Inclusion criteria
-
Peer-reviewed articles on CPB temperature management and automation.
-
Studies highlight the impact of automated systems on patient safety and procedure efficacy.
Exclusion criteria
-
Non-English articles.
-
Irrelevant topics outside the automated systems in CPB.
Selection of literature
Twelve principal articles were identified, aligning closely with our algorithm’s focus areas and the goals of enhancing CPB through automation.
Ethical considerations
Although IRB approval is not applicable, the manuscript discusses the ethical implications of automation, including human oversight, accountability, training, and alarm fatigue. Future work should include input from clinicians and ethicists.
Algorithm design
The algorithm orchestrates a series of steps designed to maintain optimal thermal conditions, based on dynamic input parameters and controlled feedback mechanisms (Figure 1).
1. Input parameters
-
Target Core Temperature: Defined by the perfusionist based on specific surgical requirements, ensuring the algorithm adapts to the varying needs of each procedure.
-
Initial Patient Temperature: Measurements are taken from multiple sites (arterial, venous, esophageal, nasopharyngeal, bladder) to establish a comprehensive baseline before initiating temperature control [7, 8].
-
Oxygen Delivery (DO2) Targets: These are set according to the metabolic demands adjusted for temperature, crucial for supporting organ function under varying thermal conditions [9, 10].
-
Cooling and Warming Rate Limits: Predefined limits ensure that temperature changes occur within safe gradients to prevent thermal shock and other temperature-related complications.
2. Automated cooling protocol
-
Initiate cooling when the target temperature is below a predefined threshold (e.g., 32 °C), controlling the rate based on the initial and target temperatures over the specified time periods [11].
-
Dynamically adjust HCU settings to modulate the temperature, ensuring efficient cooling while maintaining safe temperature gradients (ΔT) between blood and HCU.
-
Continuous monitoring of peripheral temperatures to ensure uniform cooling across all body areas [12].
-
Adapt flow rates and oxygen delivery in response to lowered metabolic demands during cooling, choosing between pH-stat and alpha-stat strategies as per institutional protocols (Figure 2) [1, 2].
 |
Figure 2 Principal flowchart. |
3. Monitoring phase
-
Real-time monitoring of arterial, venous, and core temperatures, along with the efficiency of the heat exchange system [4, 5].
-
Maintain metabolic support by balancing DO2 and consumption (VO2) and monitor CO2 production to prevent acidosis.
4. Automated rewarming protocol
-
Gradually rewarm the patient once surgical conditions permit, using controlled HCU temperature settings calculated to prevent rapid temperature increases.
-
Maintain safe temperature gradients to prevent endothelial damage and monitor for potential hyperthermic conditions.
-
System alerts to notify staff if rewarming rates exceed safe limits, ensuring immediate corrective action.
5. Final temperature adjustment and weaning
-
Stabilize core temperature within the target range prior to concluding CPB.
-
Adjust gas flow and ventilation to transition smoothly to normothermic conditions.
-
Conduct final checks of all metabolic markers to confirm patient stability before disengagement from the system.
6. Simulation protocol (preliminary validation)
A simulated environment was created using MATLAB to replicate dynamic patient responses during CPB. The algorithm was tested for accuracy, responsiveness, and redundancy during sensor dropout. Initial results showed the system maintained safe temperature gradients 97% of the time.
Algorithm integration and architecture
The system interfaces directly with standard HCUs using analog/digital communication protocols (e.g., RS232). Sensor data is continuously relayed to a central control unit. Fail-safe protocols include sensor redundancy, alarms for ∆T breaches, manual override, and logging of alerts for auditing.
Safe temperature gradient protocols
A set of predefined safe ranges for cooling and rewarming gradients is strictly maintained to minimize thermal stress and its associated risks:
-
Cooling Gradient: Maintains a maximum ΔT of 10 °C between the blood and the HCU.
-
Rewarming Gradient: Limits the ΔT to 4 °C between the core and the blood.
Deep hypothermia management
Deep hypothermia protocols are carefully managed to prevent complications associated with extreme temperature changes:
-
Gradual cooling and rewarming, closely monitoring NIRS (Near-Infrared Spectroscopy), and cerebral saturation to ensure adequate brain protection.
Oxygen and flow adjustments
DO2 levels and perfusion flows are adjusted based on temperature-dependent metabolic rates, ensuring adequate tissue perfusion across varying temperatures.
Challenges and innovations
-
The system incorporates advanced predictive modeling to dynamically adjust to patient-specific conditions and real-time changes. It addresses potential risks of over-reliance on automation by providing perfusionists with override capabilities and real-time feedback, ensuring that clinical judgment remains paramount (Figs. 3A–3H; Tables 1–5).
 |
Figure 3 Flowcharts A–H into a unified diagram that illustrates the entire temperature management process. |
Temperature management, thresholds, and nadirs from the literature.
Safe temperature gradient and threshold example for CPB.
Deep hypothermia temperature threshold example.
Temperature-based oxygen delivery and flow threshold example.
Integration of temperature, DO2, and CO2 management threshold example.
Discussion
The introduction of an automated temperature management algorithm for CPB represents a significant advancement in the field of cardiac surgery. This section discusses the benefits, limitations, and future directions for research based on an extensive review of 12 key articles [1–12]. Automated temperature management addresses the significant variability in patient outcomes attributable to manual adjustments during CPB. By dynamically adjusting temperatures based on continuous physiological feedback, the algorithm reduces the risk of cerebral hyperthermia and inflammatory responses that can lead to severe complications post-surgery [1, 2]. This proactive approach minimizes human error, enhancing patient safety and consistency in outcomes across varying clinical settings. The algorithm’s integration with advanced sensor technologies ensures reproducible and precise temperature control, essential for quality assurance in CPB practices. The standardization facilitated by such technologies not only improves the reliability of CPB but also aids in the certification of quality across healthcare institutions. Automated logging of temperature parameters provides a valuable dataset for auditing and improving CPB protocols, promoting a cycle of continuous quality improvement [3, 4]. The precision in temperature management achieved through automation directly influences organ protection and metabolic rates, crucial for optimizing patient recovery. Maintaining optimal thermal conditions reduces the incidence of postoperative complications such as acute kidney injury and enhances overall recovery [5, 6]. These improvements could lead to shorter hospital stays and better long-term health outcomes, emphasizing the clinical efficacy of automated systems [7]. While the benefits of automated temperature management in CPB are clear, some limitations and challenges necessitate further research [8]. One major concern is the algorithm’s reliance on accurate and timely data input, which can be compromised by sensor malfunctions or data integration issues. Despite these benefits, the implementation of fully automated systems introduces potential limitations that must be addressed to ensure clinical safety and acceptance. These include the risk of over-reliance on automation, which may lead to reduced vigilance or delayed clinical response during critical events. Additionally, frequent or false alarms may contribute to alarm fatigue among clinical staff, decreasing the overall responsiveness of the surgical team. Sensor malfunctions or data integration errors could result in incorrect feedback loops unless supported by robust redundancy protocols. Furthermore, the integration of such systems requires a careful balance between automation and human oversight. Maintaining manual override capabilities, providing real-time visibility of system decisions, and ensuring clear communication with perfusionists are essential safeguards. Equally important is the need for comprehensive training to ensure staff confidence in the system and the ability to intervene effectively when necessary. Future validation studies, ideally through multicenter clinical trials, are crucial to evaluate performance across diverse patient populations and surgical environments [9, 10]. Long-term monitoring will help assess the reliability, user interaction, and clinical outcomes associated with the system, refining the algorithm to better support individual patient needs while upholding safety standards [11, 12].
Conclusions
The introduction of an automated temperature management algorithm for CPB would represent a significant advancement in cardiac surgery. By reducing human error associated with manual temperature adjustments, this algorithm could enhance patient safety and contribute to more consistent surgical outcomes. It would also promote standardization across CPB procedures, ensuring a uniform approach to managing patient temperature during surgeries, which is critical for the reproducibility of successful outcomes. Despite its potential benefits, the adoption of such an advanced system poses challenges. It would require rigorous validation to ensure its reliability and effectiveness across various clinical settings. Additionally, integrating this technology into existing healthcare practices would necessitate substantial training and adaptation by medical professionals.
Funding
This research received no external funding.
Conflicts of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Data availability statement
There are no new data associated with this article.
Author contribution statement
Conceptualization, methodology, writing – original draft preparation, Y.E.D. and I.C.; original draft revision and investigation, Y.E.D. and I.C.
Ethics approval
As noted in the manuscript, IRB approval was not applicable for this study. However, we have included a section discussing the ethical considerations surrounding automation in surgical environments, including human oversight, accountability, training, and alarm fatigue.
References
- Engelman R, Baker RA, Likosky DS, et al. The Society of Thoracic Surgeons, The Society of Cardiovascular Anesthesiologists, and The American Society of ExtraCorporeal Technology: clinical practice guidelines for cardiopulmonary bypass – temperature management during cardiopulmonary bypass. J Extra Corpor Technol. 2015;47(3):145–154. [PubMed] [Google Scholar]
- Wahba A., Kunst G., De Somer F., et al. 2024 EACTS/EACTAIC/EBCP Guidelines on cardiopulmonary bypass in adult cardiac surgery. Eur J Cardiothorac Surg. 2025;67(2):ezae354. [Google Scholar]
- Saad H, Aladawy M, Temperature management in cardiac surgery. Glob Cardiol Sci Pract. 2013;2013(1):44–62. [Google Scholar]
- Mukaida H, Matsushita S, Yamamoto T, et al. Oxygen delivery-guided perfusion for the prevention of acute kidney injury: a randomized controlled trial. J Thorac Cardiovasc Surg. 2023;165(2):750–760.e5. [Google Scholar]
- Mukaida H, Matsushita S, Kuwaki K, et al. Time-dose response of oxygen delivery during cardiopulmonary bypass predicts acute kidney injury. J Thorac Cardiovasc Surg. 2019;158(2):492–499. [CrossRef] [PubMed] [Google Scholar]
- Condello I., Speziale G., Fiore F., et al. The role of continuous monitoring of venous drainage flow and integrated oxygen extraction (ERiO2) via bilateral near-infrared spectroscopy in cerebral perfusion during aortic arch surgery. Medicina (Kaunas, Lithuania). 2025;61(2):226. [Google Scholar]
- Cavaliere F., Gennari A., Martinelli L, et al. The relationship between systemic oxygen uptake and delivery during moderate hypothermic cardiopulmonary bypass: critical values and effects of vasodilation by hydralazine. Perfusion. 1995;10(5):315–321. [Google Scholar]
- Condello I., Santarpino G., Nasso G, et al. Management algorithms and artificial intelligence systems for cardiopulmonary bypass. Perfusion. 2022; 37(8):765–772. [Google Scholar]
- Kulthinee S., Warhoover M., Puis L, et al. Cardiac surgery-associated acute kidney injury in cardiopulmonary bypass: a focus on sex differences and preventive strategies. Am J Physiol Renal Physiol. 2024;327(6):F994–F1004. [Google Scholar]
- Chiba Y., Tashima Y., Ohama S., et al. Effect of nadir hematocrit during cardiopulmonary bypass on the early outcomes after surgical repair of acute type A aortic dissection. J Card Surg. 2022;37(8):2338–2347. [Google Scholar]
- Maheshwari P., Yousuf M. S., Zahid MA, et al. Acute normovolemic haemodilution to reduce allogeneic red blood cell transfusion in patients undergoing coronary artery bypass grafting: an observational study. Transfus Med. 2025;35(1):60–67. [Google Scholar]
- DeSomer F., Mulholland J. W., Bryan M. R, et al. O2 delivery and CO2 production during cardiopulmonary bypass as determinants of acute kidney injury: time for a goal-directed perfusion management? Crit Care. 2011;15(4):R192. [CrossRef] [PubMed] [Google Scholar]
Cite this article as: El Dsouki Y & Condello I. Automated temperature management during cardiopulmonary bypass: a step toward safety and precision perfusion. J Extra Corpor Technol 2025, 57, 137–146. https://doi.org/10.1051/ject/2025021.
All Tables
All Figures
 |
Figure 1 PRISMA flow chart. |
| In the text | |
 |
Figure 2 Principal flowchart. |
| In the text | |
 |
Figure 3 Flowcharts A–H into a unified diagram that illustrates the entire temperature management process. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


